“This is one of the most complex examples of parasites controlling animal behavior because it is a microbe controlling an animal – the one without the brain controls the one with the brain.” – David Hughes Penn State University
Within the cosmic battleground of Earth, an eternal symphony and conflict persist, entangling the bodies, minds, and souls of all living entities within a web of inescapable interplay.
There is no refuge from the intricate melody of life and the dark filaments of warfare that bind us, compelled by the magnetic forces of nature to partake in an unending song or engage in mutual combat.
Nothing and no one is immune.
This eternal war we can observe all around us in those victims who have lost the fight as they begin to lose their bodies and control of their minds.
Zombie ants are a perfect example of one of nature’s bizarre phenomena who lost the battle I speak.
Studies have found that they are simply infected with a fungus that takes over their bodily processes, and DNA, and corrupts them to turn them into obedient, mind-controlled servants.
Researchers found that these ants were – 50% ant and 50% fungus.
Zombies, mutants, or hybrids…
As many of you know who follow my work, I believe the same process happens to humans.
QUICK FACT: Did you know that molds/fungi have been found in 100% of autopsied human brains of people who died from Alzheimer’s disease? (Perhaps they were also taken over and their memories wiped clean by the same foreign invaders who stole the ant’s brain.)
And NO Mr. David Icke, they are NOT reptilian elites so please go back to the research desk…
Back to the ants…
A recent study showed how scientists examined the activated genes in the heads of infected zombie ants firmly attached to plants, comparing them with the heads of uninfected ants.
Their findings revealed that when ants were affixed to leaves but still displaying signs of life, only about half of the cells in their heads belonged to the ants themselves; the remainder comprised cells of the invading fungal parasite. Their cells had been combined in the blood, brain, head muscles, and fatty tissue.
During this period of zombie ant behavior, the parasitic fungus triggered a unique set of genes influencing neurotransmitters akin to serotonin, noradrenaline, and dopamine which exhibited heightened activity while the host ants manifested their peculiar zombie-like conduct.
For example, depletion of serotonin in ants is known to hinder proper foraging, and in other animals, disruption of these neurotransmitters can induce hallucinations and muscle spasms. Serotonin is involved in numerous physiological processes in ants as it is in humans such as sensorimotor skills like sleep, memory, feeding, pain, motor activity, biological rhythms, and neural development.
In other words, the fungi put the ant into a sleep-like waking state as it manipulates the behaviors in its favor by influencing these neurotransmitter systems and specific chemical processes that allow it to take over the mind.
Again, I contend that the same thing happens in humans.
As you can see, serotonin regulates and controls many functions crucial to an ant and a human’s survival and as I mentioned, this is done by our microbiota via neurotransmission.
In 2020, a study found that human neurons are like “mini computers” communicating through a root-like structure inside our bodies called dendrites.
A dendrite means “a structure of nerve cells that comprise the human brain.” The word was coined by scientists who first studied the structure of the brain, they noted its strong resemblance to trees so they named it after the Greek Dendron, meaning “tree.”
These dendrites appear to be natures, animals, mammals, and humans’ super internet signaling pathways that we all share. A type of biological internet for communication and a whole host of other mechanisms such as parasitism and natural selection.
A textbook neuron resembles a leafless tree, with extensive roots, i.e., dendrites leading to a robust, bulbous base—the body.
Electrical signals, akin to water and nutrients, ascend through dendritic roots into the body, where a hump-like structure amalgamates all information. If the sound wave/electrical stimulation is strong enough, it travels down a solitary tree trunk—the output cable, or axon—before being relayed to another neuron via bubbles containing chemical messengers or electricity.
Studies have shown that human dendrites are electrically excitable, exhibiting backpropagating action potentials and fast dendritic calcium spikes.
Dendritic processes play a fundamental role in receiving information via transducing receptors (sensory neurons) or incoming synaptic contacts (conventional neurons). In the presence of weak input signals, the neuron discards the data. Neuroscientists commonly describe single neurons as “binary” or “digital,” reflecting their tendency to either fire or remain inactive.
Through the examination of individual neurons in rodent brains, scientists have recently uncovered that dendritic trees are not merely passive cables; instead, they are highly active components that play a crucial role in a concealed layer of neural computation. Some dendritic trees, for instance, can produce electrical spikes five times larger and more frequent than the conventional firing of neurons.
Rather than recording from a living, intact human brain, the research team decided to study fresh slices of the brain’s cortex removed from patients due to epilepsy or tumors. Utilizing brain tissue from two different patient groups helped them identify signals unique to each brain disease, allowing the researchers to unravel the fundamental computations of human dendrites.
A peculiar signal quickly manifested.
Human dendrites exhibited activity, but the electrical spikes rapidly diminished as they traveled toward the cell’s surface. In contrast, a typical neural signal maintains its intensity as it travels along the output cable to its next destination.
What’s even stranger is that dendritic signals relied exclusively on calcium ions to generate electricity, a significant departure from conventional neural signaling.
The researchers concluded;
“It’s like suddenly discovering a new species that consumes carbon dioxide, rather than oxygen, to sustain its activity—except that species is part of you. ”
This is EXACTLY what I believe is happening to humans.
This species they speak of I contend are fungi/molds that can control and kill their victims, whether it be an ant, pant, or human as they see fit based on these electrical and chemical signals I speak of such as the loss of serotonin.
This loss would create a specific sound wave frequency that the fungi would use for sensing purposes to repel or magnetize their victims like what I contend happens to people who contract Alzheimer’s disease (AD). A disease with currently 50 million victims
For example, new studies have suggested that serotonin loss in humans may be a key player in cognitive decline, rather than a side-effect of Alzheimer’s disease.
As it turns out, approximately 90% of the serotonin the human body produces by our microbiota is in the gastrointestinal tract, where it regulates several bodily functions via a serotonergic pathway. Studies have found that the serotonergic pathway is modulated by gut commensal microbiota components in our gastrointestinal (GI) tract where it manages and controls the gut-brain axis.
Meaning it is our microbiome (fungi) that manages and controls (immune system) our physical and mental health.
What scientists are finding is that the microbiome has signaling mechanisms within this axis that allow it to communicate with the gut and the brain. This is called a neurotransmitter and serotonin seems to be one of the the most important mediators in microbiota–host interactions.
The serotonergic system controls the GI tract and the central nervous system (CNS) physiology. When this pathway is disrupted or corrupted, the disruption results in a wide range of pathologies that are affected thus causing a wide range of brain and intestinal diseases.
The serotonergic pathway plays a crucial role in sensorimotor function, which combines two important components: sensory input and motor output.
Sensory input to visual stimuli involves the information received through our sensory systems, including vision, hearing, smell, taste, touch, and proprioception (the sense of body position and movement).
Motor output refers to the response generated by our body in reaction to the sensory information received. Sensorimotor skills are also influenced by individual experiences and learning.
Sensorimotor skills refer to the ability to receive sensory messages from the environment and our bodies, and then generate an appropriate motor response. These skills are crucial for our daily functioning and play a fundamental role in our overall development and interaction with the world around us.
It involves the coordination and execution of movements, whether they are fine motor skills (such as writing or buttoning a shirt) or gross motor skills (such as walking or throwing a ball). These movements are the result of complex interactions between our brain, muscles, and nervous system.
Each of these sensory systems provides us with essential information about our surrounding environment and our bodies.
Sensorimotor skills are acquired and developed through a process of continuous learning and refinement from infancy through adulthood. In infancy, sensorimotor skills are foundational for the development of other cognitive and physical abilities.
Babies learn to grasp objects, visually track moving stimuli, and explore their environment through touch and taste. As they grow, they gain more control over their movements and refine their sensorimotor skills to perform more complex tasks.
Through repetition and practice, individuals refine their abilities and become more efficient in performing specific tasks. This process is known as motor learning. For example, a novice pianist may initially struggle with finger dexterity and coordination but with practice, they become more proficient in playing complex pieces.
For example, our visual system allows us to see and process visual stimuli, such as colors, shapes, and movements. Our auditory system enables us to hear and interpret sounds, while our olfactory system helps us perceive different smells.
Similarly, our taste buds allow us to experience flavors, and our sense of touch allows us to feel textures, temperatures, and pressure. Proprioception, on the other hand, provides us with information about the position and movement of our limbs and bodies in space.
Studies have indicated a correlation between motor activity and serotonergic function, and the firing rates of serotonergic neurons responding to intense visual stimuli.
In addition to the natural progression of sensorimotor skills through typical development, some individuals may experience challenges or delays in the acquisition of these skills. Sensory processing disorders, for example, can affect how individuals perceive and respond to sensory information.
Animal models propose that kainate signaling negatively influences serotonin actions in the retina, potentially impacting the regulation of the visual system. The descending projections create an inhibitory pathway referred to as the “descending inhibitory pathway,” which may have implications for disorders such as fibromyalgia, migraine, and other pain disorders, as well as the efficacy of antidepressants in treating them.
A neuron that secretes 5-HT is termed as serotonergic. It is a very important neurotransmitter in the Central Nervous System but when it becomes impaired, or damaged, it decreases in this process causing sensory processing disorders. Hence, pathology ensues along with illness and disease.
5-HT in humans is extensively present in various bodily systems, such as the nervous, gastrointestinal, and cardiovascular systems. It influences a broad range of physiological and pathological conditions, including pain, sleep regulation, aggression, feeding, anxiety, and depression.
The disturbance of 5-HT signaling in various pain states has been observed in both basic research and clinical studies, suggesting a potential explanation for certain diffuse pain conditions. In certain neuropathic pain models, the baseline level of 5-HT in the spinal cord was found to be reduced.
Researchers have found that the disruption of 5-HT neurotransmission contributes to the decline in cognitive processes associated with aging, Alzheimer’s disease (AD), and various neuropathologies, including schizophrenia, stress, mood disorders, and depression. Also, people with autoimmune disorders like AIDS and similar diseases have significantly lower 5-HT and an increased rate of infections.
Multiple studies have affirmed the pathophysiological importance of the 5-HT system in AD, with several drugs enhancing 5-HT neurotransmission proving effective in addressing AD-related cognitive and behavioral deficits.
5-HT receptors, 5-hydroxytryptamine receptors, or serotonin receptors, are a group of G protein-coupled receptor and ligand-gated ion channels found in the central and peripheral nervous systems. They mediate both excitatory and inhibitory neurotransmission.
As I explained in my previous essay, recent studies suggest that vibrations caused by sound waves directly affect the ion channels in fungal cells, resulting in electrical activity. Other hypotheses propose that sound-induced electrical responses in fungi are linked to their role in communication, growth, or defense mechanisms.
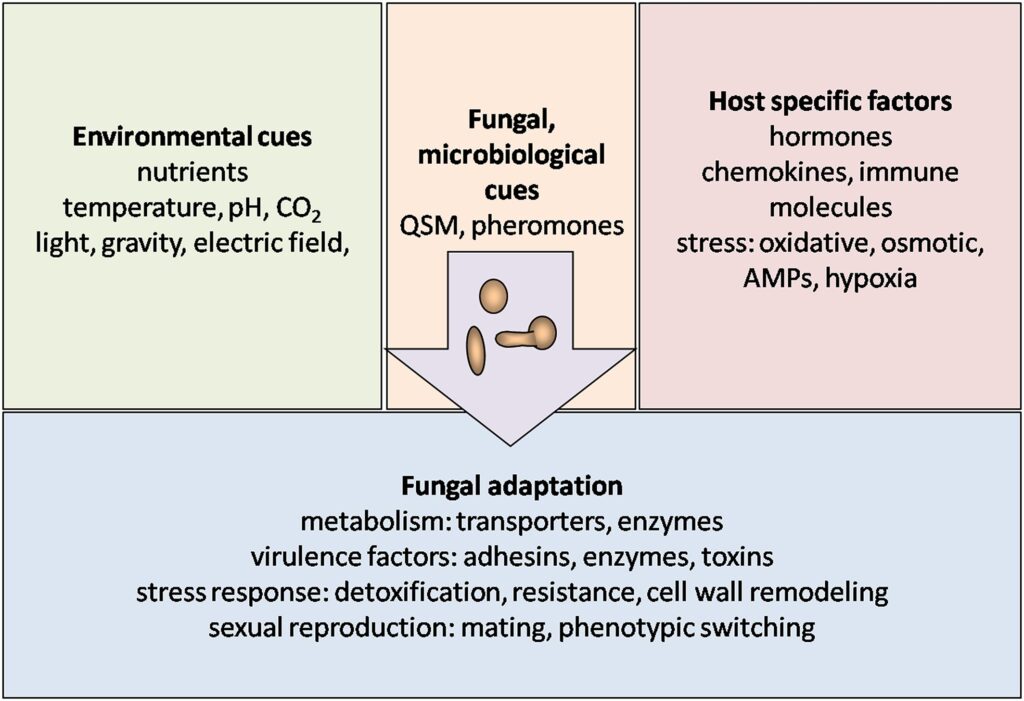
As it relates to nature, scientists have identified that the main jobs of fungi are breaking down organic matter, and processing nutrients and chemicals in a commensal, symbiotic, or pathogenic relationship with its host. For fungi to thrive within a host, they must navigate a dynamic and often challenging environment, necessitating the capacity to perceive and understand their surroundings.
Under typical circumstances, predisposing host factors, like immune suppression, play a crucial role in the survival and propagation of pathogens within mammalian hosts. Once inside the host, these pathogens must contend with the host’s microbiota for essential nutrients. For opportunistic pathogens, breaches in the normal physiological barrier, whether in mammals or plants, serve as entry points.
Fungi have developed various virulence mechanisms to elude the host’s immune system, a topic thoroughly explored elsewhere (Collette and Lorenz, 2011). Sensing these external cues is necessary to adjust fungal morphology, metabolism, mating, and virulence. Furthermore, extensive reviews have delved into how fungi sense environmental cues such as nutrients, gasses, light, and stress (Bahn et al., 2007).
Fungal infections have become a significant medical challenge, and a team of researchers at Baylor College of Medicine has made a significant breakthrough in studying the short-term effects of fungal infection in the brain. According to a study published in the journal Nature Communications, the researchers discovered that Candida albicans, a common yeast and type of fungus, can cross the blood-brain barrier and trigger an inflammatory response in mice.
This response led to the formation of granuloma-type structures and temporary mild memory impairments in mice.
Researchers injected C. albicans into mice’s bloodstream and discovered that the yeast can cross the blood-brain barrier, triggering the activation of microglia cells in the brain. The microglia cells became highly active, consuming and digesting the yeast, while also producing molecules that caused an inflammatory response.
This led to the formation of a granule-type structure called fungus-induced glial granuloma (FIGG). The mice infected with the yeast showed impaired spatial memory, which improved once the infection cleared. Although the yeast infection cleared in about 10 days, the microglia cells remained active and the FIGGs persisted for at least 21 days.
These amyloid molecules are typically associated with Alzheimer’s disease.
“These findings suggest that the role fungi play in human illness potentially goes well beyond allergic airway disease or sepsis,” according to Dr. David B. Corry, professor of medicine-immunology, allergy and rheumatology and Fulbright Endowed Chair in Pathology at Baylor College of Medicine.
“The results prompted us to consider the possibility that in some cases, fungi also could be involved in the development of chronic neurodegenerative disorders, such as Alzheimer’s, Parkinson’s and multiple sclerosis. We are currently exploring this possibility,” Dr. Cory said.
Since they do not have eyes and a nose to successfully adapt, fungi must be attuned to external environmental and biochemical factors to process this information to respond and identify unique host-specific elements. They do this by sensing and reacting to the host’s temperature, pH, gasses, nutrients such as sugars, amino acids, nitrogen, and other trace elements along with serotonin, which are all essential for the growth and viability of fungal symbioses or pathogeneses in every environment.
Given that the fungal cell wall maintains constant contact with its surroundings, the expression of receptors, such as pheromone receptors, on the cell wall surface becomes crucial.
Pheromone receptors are proteins that are sensitive to pheromones, which are chemical signals that organisms release to communicate with each other. These signals play a crucial role in various biological processes, including mating, territory marking, and social organization. Pheromone receptors are found in a wide range of organisms, from bacteria and fungi to insects and mammals.
In 1961, researchers noted that some “chemical messengers” act within an individual (e.g., hormones and “other excitatory substances” such as CO2), whereas others (i.e., “pheromones”) act between individuals via ingestion, absorption, or sensory receptors. However, fast forward to 2023, various studies have not revealed conclusively if humans create pheromones on their own.
Considering the chance that we do not create them on our own, the question arises: where do they come from?”
Pheromones in humans may be present in bodily secretions such as urine, semen or vaginal secretions, breast milk, and potentially also saliva and breath, yet most attention thus far has been directed toward axillary sweat. However, studies suggest that we can detect each other through unique smells produced by signaler pheromones
In fungi, pheromone receptors are particularly important for sexual reproduction. They are eukaryotic organisms that reproduce both sexually and asexually.
During sexual reproduction, fungi use pheromones to signal their mating compatibility with other individuals of the same species.
This process is essential for the fusion of specialized sexual structures called gametangia, ultimately leading to the formation of new genetically diverse individuals.
Here is an image explaining this biological process:
The typical lifecycle of fungi involves the following steps:
1. Pheromone production: Fungi release pheromones into their environment. These pheromones act as signaling molecules, indicating the presence and mating compatibility of the releasing individual
2. Pheromone reception: Potential mating partners have pheromone receptors on their cell surfaces. These receptors are specific to the type of pheromones produced by compatible mating partners.
3. Chemotropism: The receiving fungal cells respond to the pheromones by growing towards the source of the pheromone (a process known as chemotropism). This directional growth helps mating partners to come into proximity.
4. Cell fusion (plasmogamy): Once the compatible cells come into contact, they undergo cell fusion or plasmogamy. This fusion of cytoplasmic contents is a crucial step in sexual reproduction.
5. Formation of sexual structures: Following plasmogamy, specialized structures such as a zygote or a dikaryotic mycelium are formed, depending on the fungal species.
6. Completion of sexual reproduction: The sexual structures eventually lead to the formation of spores or other structures that can disperse and give rise to new individuals.
The entire process is tightly regulated by the interaction between pheromones and their corresponding receptors. The specificity of these interactions ensures that mating occurs only between compatible individuals of the same fungal species. The study of pheromone signaling in fungi has provided valuable insights into the molecular mechanisms underlying sexual reproduction in eukaryotic organisms.
These pheromone receptors not only facilitate chemotropism for mating but also serve other essential roles. These chemicals in the body are “electrically charged” — when they have an electrical charge, they are called ions. ‘
The term “ion” finds its origin in the Greek language, specifically derived from the neuter present participle of “ienai” (Greek: ἰέναι), which translates to “to go.” In the realm of ions, a cation is associated with downward movement (Greek: κάτω pronounced kato, meaning “down”), while an anion is linked to upward movement (Greek: ano ἄνω, meaning “up”).
It is this ion channel that I believe is the main method that fungi use to exploit and corrupt once the negative charge ions become too imbalanced
An ion, as introduced by English physicist and chemist Michael Faraday in 1834, is a term used to describe a species that travels from one electrode to another through an aqueous medium. This concept was developed after a suggestion by the English polymath William Whewell. To understand the concept of an ion, it is necessary to delve into the world of chemistry and electricity.
In the realm of electrochemistry, ions play a crucial role. An ion is an atom or a molecule that has gained or lost one or more electrons, resulting in a net positive or negative charge. These charged particles are formed when an atom gains or loses electrons to achieve a stable electronic configuration.
The operation of batteries also relies on ion migration.
In a typical battery, chemical reactions occur at the electrode surfaces, resulting in the generation of a voltage. This voltage drives the migration of ions between the electrodes, allowing for the transfer of charge and the production of electrical energy.
During discharge, positive ions move from the anode to the cathode, while negative ions move in the opposite direction. This ion migration enables the flow of electrons through an external circuit, producing an electric current.
The term “ion” was coined by Faraday to describe the movement of these charged species during electrolysis. Electrolysis is a process that utilizes an electric current to drive a non-spontaneous chemical reaction. It involves the decomposition of an electrolyte, a substance that conducts electricity when dissolved in a solvent, typically water.
During electrolysis, an external electric current is applied to an electrolytic cell, which consists of two electrodes, an anode (positive electrode) and a cathode (negative electrode), immersed in an electrolyte solution. When the electric current flows through the cell, ions are attracted to the respective electrodes based on their charge.
The movement of ions from one electrode to another through the aqueous medium is what Faraday referred to as an ion catation. The term “catation” is derived from the Greek word “kation,” meaning “to go down.” This reflects the movement of positively charged ions, known as cations, towards the cathode.
On the other hand, negatively charged ions, known as anions, migrate towards the anode during electrolysis. These anions are formed when an atom gains one or more electrons, resulting in a negatively charged species. The migration of anions is often referred to as anion catation.
Anion (−) and cation (+) indicate the net electric charge on an ion. An ion that has more electrons than protons, giving it a net negative charge, is named an anion, and a minus indication “Anion (−)” indicates the negative charge. With a cation it is just the opposite: it has fewer electrons than protons, giving it a net positive charge, hence the indication “Cation (+)”.
In addition to their roles in electrochemical processes, ions are also essential for maintaining the balance of charges in various biological systems. In living organisms, ions such as sodium (Na+), potassium (K+), calcium (Ca2+), and chloride (Cl-) play vital roles in nerve conduction, muscle contraction, and maintaining osmotic balance.
The movement of ions across cell membranes through specialized ion channels allows for the transmission of electrical signals and the regulation of cellular functions.
HOW FUNGI USE SOUND WAVES AND FREQUENCIES CONTROL THE WORLD
Studies have shown that fungi are extremely polarized organisms that constantly produce internal electrical currents and fields that are generated by hyphae. Its growth requires a constant supply of proteins and lipids to the hyphal tip.
Researchers have proven that the individual hyphae of the filamentous fungi constantly perform cellular “monologue” and cell-to-cell dialog using signal oscillations to acquire or magnetically attract the nutrients it requires to grow and thrive within the host.
For example, studies have found how psilocybin reduces low-frequency oscillatory power in users’ brains, increases overall firing rates, and desynchronizes local neural activity. It indicates experiences correlate with the lagged phase synchronization of delta oscillations. Schizophrenia, which mimics symptomatically the psychotic effects of psilocybin, is associated with diffuse delta rhythms.
These oscillations create electricity which is one of the key factors shaping their growth and development. The hyphae become polarized and entrained as the branching of mycelium is induced by electric field frequency, which it uses to communicate and transport the raw human materials within the blood and central nervous system.
The electrical current helps fungi with the translocation of resources it gathers within the host using magnets and hydraulic pressure.
Recent studies have explored the potential of utilizing frequency-specific sounds as a viable substitute for chemical fungicides in combatting plant diseases. The research findings indicated that high frequencies possess the ability to impede mycelium growth, akin to the impact of high-pitched noises causing deafness.
Further investigation revealed noticeable morphological changes in the mycelium, providing insights into the mechanism. This suggests that certain sound wave frequencies can induce stressful growth conditions, presenting a sustainable approach to combating pathogenic fungal pathogens.
As signaling and metabolism in organisms are controlled by a precise ionic gradient across membranes, the disruption of this gradient contributes to cell death. This is a common mechanism exploited by natural and artificial biocides, including the ion channels gramicidin and amphotericin (a fungicide).
What is interesting and helps prove my theory is that researchers have recently found that 5-HT is a strong inhibitor of fungal growth.
So, not only are ion channels used to treat people with antifungal medicines, but supplementing with 5-HT in the serotonergic pathways can stop fungi from growing in the body and brain. As I mentioned, the serotonergic pathway is modulated by the gut commensal where 5-HT is biosynthesized with L-tryptophan (Trp) derived from our diets.
As I mentioned, the loss of serotonin or 5-HT causes a condition known as sensory processing disorder (SPD), which interferes with the typical processing of sensory information (stimuli) in the brain. This involves the processing of what you see, hear, smell, taste, or touch.
SPD may impact all senses or just one, resulting in heightened sensitivity to stimuli compared to the general population. These disorders can result in hypersensitivity or hyposensitivity to certain sensory stimuli, leading to difficulties in regulating behavior and responding appropriately to the environment.
For example, ants normally do not venture out solo and climb a big tree to nowhere destined to become its aerial deathbed.
Once the ant victim clamps down on the leaf, the parasitic fungus triggers a series of genes responsible for the degradation of the ants’ jaw muscles, resulting in the lockjaw effect. Simultaneously, it activates genes that suppress the ant’s immune system, facilitating the unimpeded growth and proliferation of fungal cells throughout the ant’s head tissues.
As the ants cease their struggle and succumb to the fungus, a staggering 75% of the cells in their heads transform into fungal cells.
The ant is no longer an ant but a parasitic fungus that has taken over an ant carcass, thus becoming a “zombie ant.”
During this period, numerous genes in the fungal genome related to ant host digestion, cell growth, and reproduction shift into high gear, marking the fungus’s transition to a rapid growth phase for the development of its reproductive stalk so it can shoot spores to infect other ants.
In another study from 2019, researchers found that at the moment of behavioral manipulation by the fungus, the host’s brain is not invaded by the fungus.
Instead, it invades other areas of the ant’s muscle tissue making them a co-pilot.
They discovered that despite not being invaded by the parasite, the brains of manipulated ants are notably different, showing alterations in neuromodulatory substances, signs of neurodegeneration, changes in energy use, and antioxidant compounds that signal stress reactions by the host.
I have often wondered if fungi/molds can do this to ants and other insects, why not humans?
After all, these tiny but deadly creatures have been around for millions of years and are well known for their industrious nature and strong social organization.
This so-called co-pilot stage may be what we witness in individuals with early-stage Alzheimer’s disease where the fungal cells eliminate and displace the human cells like it does the ant. Thus causing various pathologies.
At this stage, we call it dementia.
Ergothionine, a fungal-derived compound with known neuronal cytoprotection functions was found to be highly elevated in zombie ant brains suggesting the fungus, which does not invade the central nervous system, is preserving the brain.
Ergothioneine is a naturally occurring amino acid compound that is produced in relatively few organisms, notably actinomycetota, cyanobacteria, and certain fungi. Ergothioneine was first discovered in 1909 and named after the ergot fungus from which it was first purified.
The researchers found thousands of unique chemicals, most of them completely unknown. This, according to Hughes, is not surprising, since little previous work has mined these fungi for the chemicals they produce.
But what did stand out were two known neuromodulators, guanobutyric acid (GBA) and sphingosine. These both have been reported to be involved in neurological disorders and were enriched when the fungus was grown in the presence of the brains of its target species.
“There is no single compound that is produced that results in the exquisite control of ant behavior we observe,” de Bekker said. “Rather, it is a mixture of different chemicals that we assume act in synergy.
“But whatever the precise blend and tempo of chemical secretion,” she said, “it is impressive that these fungi seem to ‘know’ when they are beside the brain of their regular host and behave accordingly.”
Noted Hughes, “This is one of the most complex examples of parasites controlling animal behavior because it is a microbe controlling an animal — the one without the brain controls the one with the brain.
By employing metabolomics and controlled laboratory infections, we can now begin to understand how the fungi pull off this impressive trick.”
GNOSTIC WARRIOR CONCLUSION:
The research clearly shows that fungi can zombify an ant via a multi-prong approach. Meaning, there is not one specific method or neurotransmitter that the fungi use to manipulate its host.
Hence, it is hacking multiple bodily and mental processes to achieve its aim – Total Control.
To study this phenomenon, scientists use data sets called multiomics or “panomics” or “pan-omics” as a biological analysis approach to analyze complex biological big data to discover novel associations between biological entities, pinpoint relevant biomarkers, and build elaborate markers of disease and physiology.
The meaning of multiomics is to study life in a synergetic way using data sets with multiple “omes“, like the genome, proteome, transcriptome, epigenome, metabolome, and microbiome. That is essentially what I’m doing in the creation of this essay but also using a multidisciplinary scientific and logical approach to my theory.
A 2023 “multiomic” study found the dysregulation of neurotransmitter levels and neuronal signaling. The researchers believe this alteration or corruption occurs during infection, which immediately triggers;
1 – differential expression of neurotransmitter synthesis and receptor genes
2 – altered abundance of metabolites and neurotransmitters (or their precursors) with known behavioral effects in ants and other insects, and
3 – possible suppression of a connected immunity pathway. We additionally report signals for metabolic activity during manipulation related to primary metabolism, detoxification, and anti-stress protectants.
The researchers concluded;
“Taken together, these findings suggest that host manipulation is likely a multi-faceted phenomenon, with key processes changing at multiple levels of molecular organization.”
What is important to understand is that these alterations in the body lead to changes in animal host behavior mostly referred to as manipulations, preceding a fatal change in behavior. For example, the infected zombie ants, they began to stop communicating with their fellow ants as they then left their nest and normal foraging trails venturing solo into the forest which is not natural.
In our human society, we can witness similar traits among the mentally ill or people who have a disease and the alcoholic and drug addicts of our world. Their bodies and brains have been altered or corrupted
Scientists speculate that the diverse alterations observed in hosts might serve as exploitable traits for fungal parasites. This strategy allows the parasites to exploit host behaviors and symptoms without the need for costly host rewiring.
They believe that these various changes may represent behaviors that can be easily coopted for manipulation by fungal parasites.
The parasitical fungi may be taking advantage of existing host processes and symptoms without relying on costly mechanisms to “rewire” their hosts.
The same process I believe occurs in humans with similar zombie-like traits.
The long list of diseases and addictions killing people worldwide makes me ponder if these same fungi are manipulating our thoughts and behaviors, which seems to be affecting almost everyone alive.
It is the alcoholic who cannot stop drinking the very poison that is killing them. (Globally an estimated 237 million men and 46 million women suffer from alcohol-use disorders. – WHO)
It is the obese person who for the life of them, cannot stop eating junk that will cause them to have a heart attack and die. (Worldwide, more than 1 billion people have obesity—650 million adults, 340 million adolescents, and 39 million children. – WHO)
Millions of people losing control of their bodies as they lose their minds.
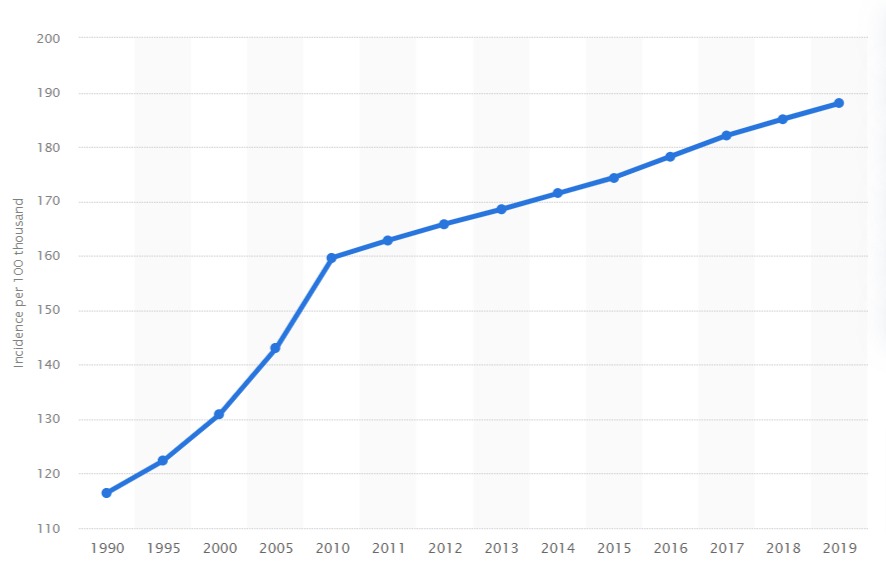
Annual incidence of Alzheimer’s disease and other dementias in Europe from 1990 to 2019(per 100,000)
The reengineering of potentially billions of people around the world within a Fungi Deep State.
Hundreds of millions of people have lost the ability to think or behave like a normal human being.
A globe covered by human fungal mutants or who we would call addicts and the mentally ill.
A defacto death sentence for the host but food and a playground for the very fungi who made it all happen – The Zombie Apocalypse.
SOURCES:
https://www.sciencedaily.com/releases/2014/08/140825142124.htm
https://www.sciencedirect.com/science/article/pii/S1568163721003032
https://www.frontiersin.org/articles/10.3389/fendo.2021.748254/full
https://www.bcm.edu/news/fungi-cause-brain-infection-in-mice
https://www.ncbi.nlm.nih.gov/pmc/articles/PMC6942873/
https://www.sciencedirect.com/science/article/abs/pii/S0301008212001086
https://pubmed.ncbi.nlm.nih.gov/27155351/
https://onlinelibrary.wiley.com/doi/10.1111/cmi.12610
https://www.ncbi.nlm.nih.gov/pmc/articles/PMC3010239/#:~:text=5%2DHT%20was%20found%20to,hyphal%20elongation%20of%20Aspergillus%20spp.
https://www.nature.com/articles/s41598-023-40065-0

Moe is the founder of GnosticWarrior.com. He is a father, husband, author, martial arts black belt, and an expert in Gnosticism, the occult, and esotericism.